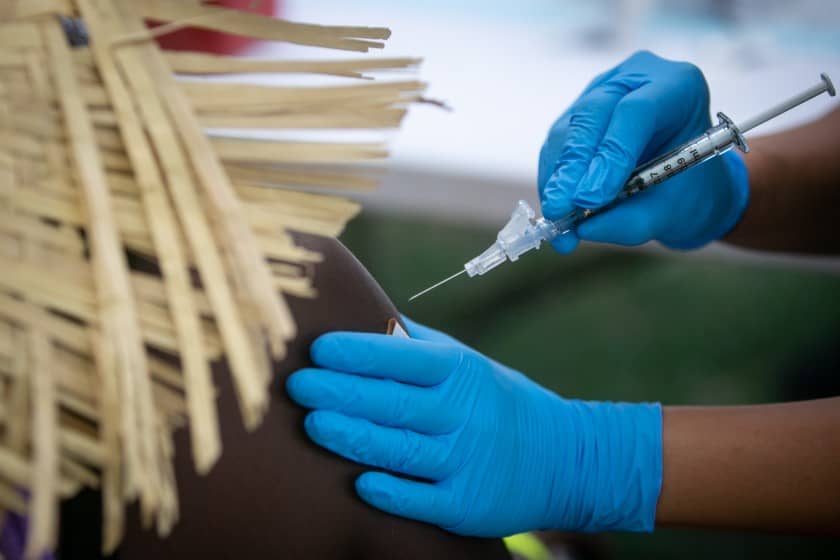
Following days of lengthy debate among vaccine experts, booster shots of the Pfizer/BioNTech Covid-19 vaccine can now be officially administered to some adults in the United States.

Early Friday morning, US Centers for Disease Control and Prevention Director Dr. Rochelle Walensky diverged from the agency’s independent vaccine advisers to recommend boosters for a broader group of people — those ages 18 to 64 who are at increased risk of Covid-19 because of their workplaces or institutional settings — in addition to older adults, long-term care facility residents and some people with underlying health conditions.
The CDC recommendation includes the recommendations made by its Advisory Committee on Immunization Practices on Thursday, when it voted to recommend boosters for people age 65 and older and residents of long-term care facilities who received the Pfizer/BioNTech Covid-19 vaccine series at least 6 months ago, and people ages 50 to 64 with underlying medical conditions.

The CDC also endorsed the vaccine advisers’ recommendation that people ages 18 to 49 with underlying medical conditions may receive a booster based on their individual benefits and risks.
However, ACIP members voted 9-6 against recommending boosters for people ages 18 to 64 who were at greater risk because of their occupational or institutional settings, such as health care workers, caregivers for frail or immunocompromised people, people in homeless shelters and people in correctional facilities.
Walensky’s recommendation aligned with the US Food and Drug Administration; on Wednesday, it gave emergency use authorization for a booster dose of Pfizer’s Covid-19 vaccine in people 65 and older, people at high risk of severe disease and people whose jobs put them at risk of infection.
“As CDC Director, it is my job to recognize where our actions can have the greatest impact. At CDC, we are tasked with analyzing complex, often imperfect data to make concrete recommendations that optimize health. In a pandemic, even with uncertainty, we must take actions that we anticipate will do the greatest good,” Walensky said in the statement.
“I believe we can best serve the nation’s public health needs by providing booster doses for the elderly, those in long-term care facilities, people with underlying medical conditions, and for adults at high risk of disease from occupational and institutional exposures to COVID-19. This aligns with the FDA’s booster authorization and makes these groups eligible for a booster shot. Today, ACIP only reviewed data for the Pfizer-BioNTech vaccine. We will address, with the same sense of urgency, recommendations for the Moderna and J&J vaccines as soon as those data are available.”
Debates about boosters ramped up in August when US health officials announced the United States would offer them to people who had received mRNA vaccines about eight months earlier, pending signoff from the FDA and CDC. However, data about the safety and efficacy of boosters doses was limited.
Pfizer had asked the FDA for full approval of a booster dose for everyone 16 and older six months after they were fully immunized with two shots. The company argued that it had enough evidence that immunity starts to wane after six months and that giving a booster restores the immunity safely.
More data on booster doses is being generated in the United States and Israel, in particular, and eligibility for boosters may open up in the future.
Moderna has asked the FDA to authorize booster shots for its vaccine, but the FDA so far has only considered Pfizer’s bid. Johnson & Johnson released partial data this week that it said showed a booster dose greatly raised immunity, but the company has not yet applied with the FDA to consider a booster vaccine dose.
Advertise with the mоѕt vіѕіtеd nеwѕ ѕіtе іn Antigua!
We offer fully customizable and flexible digital marketing packages.
Contact us at [email protected]