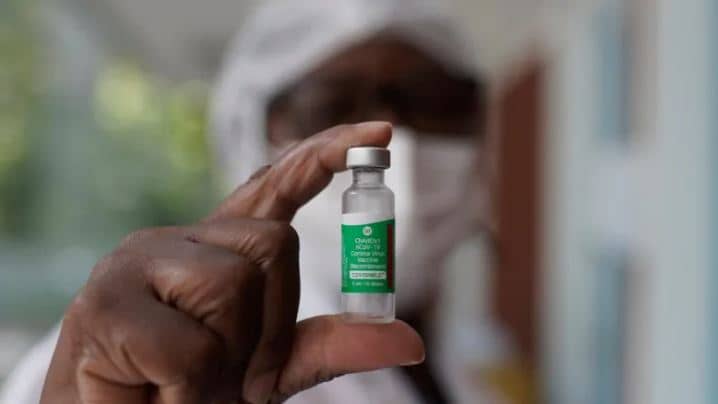
Denmark has suspended the AstraZeneca vaccine after cases of blood clotting, despite the EU ruling that a death in Austria had not been caused by the jab.
Denmark’s health minister said authorities were probing whether there was a link between having the jab and blood clots, after several cases and one death.
Magnus Heunicke said: ‘At present it can not be concluded whether there is a connection. We are acting early, it must be thoroughly investigated.’

Denmark joins five other countries – Austria, Estonia, Lithuania, Luxembourg and Latvia – who suspended the vaccine after a 49-year-old Austrian woman died of ‘severe blood coagulation problems.’
The EU’s medicines watchdog yesterday announced that it had found no link between the AstraZeneca vaccine and the woman’s death.
Trials of the vaccine showed adverse reactions were short-lived for the most part and blood clotting issue were not reported. Picture: File image
‘There is currently no indication that vaccination has caused these conditions, which are not listed as side effects with this vaccine,’ the EMA regulator said.
As well as the woman’s death, another three cases of blood clotting issues had been reported in Austria from people who had taken the vaccine.
One of those patients, a 35-year-old woman, developed a pulmonary embolism – a blockage of an artery on the lung – but is now recovering.
The EMA said that the batch which had been singled-out in Austria, labelled ABV5300, comprised one million doses and had been delivered to 17 EU countries.

The EMA said: ‘Although a quality defect is considered unlikely at this stage, the batch quality is being investigated.’
It said the Pharmacovigilance Risk Assessment Committee (PRAC) were investigating the thrombosis conditions linked to the same batch as well as all other reports of thrombosis and related blood clotting conditions reported post-vaccination.
‘The information available so far indicates that the number of thromboembolic events in vaccinated people is no higher than that seen in the general population,’ it said.
The EMA said as of Tuesday, there had been 22 reports of such conditions among the three million people to receive the AstraZeneca jab in the European Economic Area.
It said further information would be released as the investigation progresses.
EU regulators on January 30 approved the AstraZeneca vaccine, saying it was effective and safe to use.
Adverse reactions seen in trials were short-lived for the most part and blood clotting issues were not reported.
The investigation comes after a humiliating U-turn by EU leaders who backed the vaccine after claiming it was ineffective at the height of their row with the vaccine-maker and Britain in January.
During the ongoing row, the EU has furiously blamed Britain for its woeful vaccine rollout, blocked doses from leaving to Australia and begged the USA to give them its surplus doses.
Last week, Italy stood at the European vanguard as it embargoed 250,000 doses which were destined for Sydney.
Advertise with the mоѕt vіѕіtеd nеwѕ ѕіtе іn Antigua!
We offer fully customizable and flexible digital marketing packages.
Contact us at [email protected]
















My issue is that Caribbean health officials are not testing and and creating their own data. They rely solely on the developed countries for information. Why don’t we do blood test before the vaccines and then if there are major side effects, they run the before and after blood test to see if anything developed after the vaccine. We need to run our own test based on our climatic condition, ethnicity etc our livestyle is different than the developed world
Comments are closed.